Why is hydroxide a bad leaving group?
 James Craig
James Craig 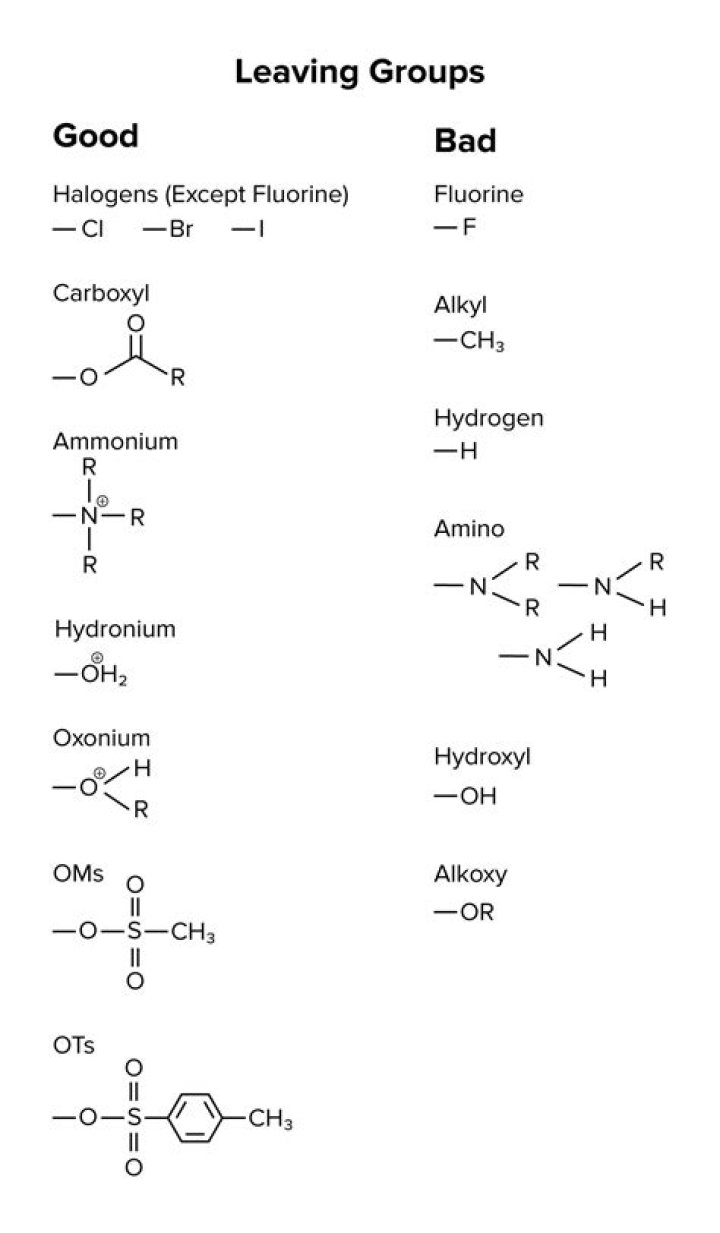
Keeping this in consideration, what makes a bad leaving group?
In order for a leaving group to leave, it must be able to accept electrons. A strong bases wants to donate electrons; therefore, the leaving group must be a weak base. This is because an increase in electronegativity results in a species that wants to hold onto its electrons rather than donate them.
Additionally, is hydroxide a strong or weak nucleophile? A good base is usually a good nucleophile. So, strong bases — substances with negatively charged O, N, and C atoms — are strong nucleophiles. Examples are: RO?, OH?, RLi, RC≡C:?, and NH2?. Some strong bases are poor nucleophiles because of steric hindrance.
Hereof, is OH or h2o a better leaving group?
The Conjugate Acid Is Always A Better Leaving Group
Its conjugate base is HO(–). When we add acid, water becomes H3O(+), which has a pKa of –1.7. It's a much stronger acid, in other words, and therefore its conjugate base (water, H2O) is much weaker.
Is methanol a good leaving group?
Good for Sn1, not the best for Sn2. 4) Leaving group: Br -, a very good leaving group. Decision: Methanol is a polar protic solvent, which is good for a Sn1 reaction.
Related Question Answers
IS F a good leaving group?
The trend is pretty clear – in general, the weaker the base, the better the leaving group. Fluorine tends to be a very poor leaving group for SN1/SN2/E1/E2 reactions. In Org 2, you may see some examples where F can act as a leaving group when it is attached to a carbonyl carbon or an aromatic ring.Is I or Br a better leaving group?
Now as we know, a strong acid will have a weak conjugate base. Since HI is the strongest acid, Iodide ion will be the weakest conjugate base within the group. So the order for increasing basic character is I<Br<Cl<F. Hence it will be a better leaving group than flouride ion.What is a good leaving group?
Good leaving groups are weak bases. They're happy and stable on their own. Some examples of weak bases: halide ions (I-, Br-, Cl-) water (OH2), and sulfonates such as p-toluenesulfonate (OTs) and methanesulfonate (OMs). The weaker the base, the better the leaving group.Why is iodine a better leaving group than chlorine?
Iodine is a better leaving group than other halogen atoms due to its larger size. Due to larger size, charge density decreases and it becomes stable. So, its a better leaving group.Why does the leaving group leave in sn1?
Effects of Leaving GroupA good leaving group wants to leave so it breaks the C-Leaving Group bond faster. Once the bond breaks, the carbocation is formed and the faster the carbocation is formed, the faster the nucleophile can come in and the faster the reaction will be completed.
What are some strong nucleophiles?
Strong nucleophiles:| VERY Good nucleophiles | HS–, I–, RS– |
|---|---|
| Good nucleophiles | Br–, HO–, RO–, CN–, N3– |
| Fair nucleophiles | NH3, Cl–, F–, RCO2– |
| Weak nucleophiles | H2O, ROH |
| VERY weak nucleophiles | RCO2H |
How do you make a good leaving group Oh?
There are three main ways to do this.- Turn it into an alkyl halide through the use of PBr3 or SOCl2.
- Turn it into an alkyl sulfonate using tosyl chloride (TsCl) or mesyl chloride (MsCl).
- Add acid.
Is h2o a better nucleophile than OH?
Notice that when oxygen is part of the hydroxide ion, it bears a negative charge, and when it is part of a water molecule, it is neutral. The O of -OH is a better nucleophile than the O of H 2O, and results in a faster reaction rate.Why is water a good leaving group?
Water is the conjugate base of hydronium ion, the strongest acid that can exist in water. Thus water is a weak base. It has little tendency to share its electron pairs with another species. Water in the form of hydronium ion is an excellent leaving group.Why is water a better nucleophile than hydroxide?
Water has no charge hence it does not bond with compounds. Hydroxide ions are negative and using the knowledge of opposite attract, this shows hydroxide ions are better nucleophile than water. The more mass the halogen have, the more electron shielding thus less attraction.Are Alkoxides good leaving groups?
Hydroxide and alkoxide ions are not good leaving groups; however, they can be activated by means of Lewis or Brønsted acids. Triflate, tosylate and mesylate are the anions of strong acids. The weak conjugate bases are poor nucleophiles. Nucleophilicity increases in parallel with the base strength.Are ethers good leaving groups?
The leaving groups of alcohols and ethers (HO-, RO-) are much stronger bases than the leav- ing group of an alkyl halide. Because they are stronger bases, they are poorer leaving groups and, therefore, are harder to displace.Which is the weakest base?
Ammonia is a typical weak base. Ammonia itself obviously doesn't contain hydroxide ions, but it reacts with water to produce ammonium ions and hydroxide ions. However, the reaction is reversible, and at any one time about 99% of the ammonia is still present as ammonia molecules.How do you tell if a nucleophile is strong or weak?
Nucleophilicity is measured by comparing reaction rates; the faster the reaction, the better (or, “stronger”) the nucleophile.How can you tell if a nucleophile is strong or weak?
- So the E2 and SN2 reactions require “stronger” nucleophiles/bases than the SN1 and E1 reactions.
- Strong nucleophiles generally bear a negative charge, such as RO(-), (-)CN, and (-)SR.
- Weak nucleophiles are neutral and don't bear a charge.
- Example 1 uses NaCN (a strong nucleophile).
Is s a strong base?
Hydroxide ion isn't hydrolyzing water to create hydroxide; the below equation is non-sensical. Therefore, sulfide ion is the stronger base.Why is ammonia a weak alkali?
The aqueous solution of ammonia is weak alkali because ammonia produces OH- ions in it's water solution. - Initially ammonia doesn't have any kind of OH- or hydroxyl ions. - Due to the presence of OH- or hydroxyl ions in the solution,the aqueous solution of ammonia is considered as a weak alkali.What is the strongest base?
Sodium hydroxide is the strongest base as it completely dissociates to give sodium ions and hydroxide ions. These hydroxide ions reacts with hydrogen ions from the acid and completely ionises the hydrogen ions.Is NaOH a weak nucleophile?
Take a species like NaOH. It's both a strong base and a good nucleophile. When it's forming a bond to hydrogen (in an elimination reaction, for instance), we say it's acting as a base. Similarly, when it's forming a bond to carbon (as in a substitution reaction) we say it's acting as a nucleophile.Is Hi a strong acid?
HCl, HBr, and HI are all strong acids, whereas HF is a weak acid. The acid strength increases as the experimental pKa values decrease in the following order: Hydrochloric acid: Hydrochloric acid is a clear, colorless solution of hydrogen chloride (HCl) in water.Which hydroxide is a weak base?
For example, acetic acid (HC2H3O2) and oxalic acid (H2C2O4) are weak acids, while iron hydroxide, Fe(OH)3, and ammonium hydroxide, NH4OH (which is actually just ammonia, NH3, dissolved in water), are examples of weak bases.Is sn1 or sn2 faster?
We were studying about nucleophilic substitution reactions. My professor said that in general SN1 reactions are faster than SN2 reactions. In this case, what I think is that the rate will depend on our reagent, leaving group, solvent, etc and in some cases SN1 will be faster while in some others SN2.What is the best leaving group for sn1?
Effects of Nucleophile- Reaction proceeds via SN1 because a tertiary carbocation was formed, the solvent is polar protic and Br- is a good leaving group.
- This reaction occurs via SN1 because Cl- is a good leaving group and the solvent is polar protic.
Is ammonia a good leaving group?
The difference between the eliminations of alcohols and amines in acidic solution is the poorer leaving group ability of ammonia than that of water (remember, ammonia is a stronger base; therefore a poorer leaving group.)Is THF Protic or aprotic?
Among the most important are whether the solvents are polar or non-polar, and whether they are protic or aprotic.Solvent Polarity.
| Solvent | Boiling Point, Celsius | Dielectric Constant |
|---|---|---|
| acetic acid, CH3COOH | 118 | 6 |
| POLAR APROTIC SOLVENTS | ||
| dichloromethane, CH2Cl2 | 40 | 9.1 |
| tetrahydrofuran (THF), cyc-(CH2)4O | 66 | 7.5 |