What is a pharmacokinetic study?
 Rachel Ellis
Rachel Ellis 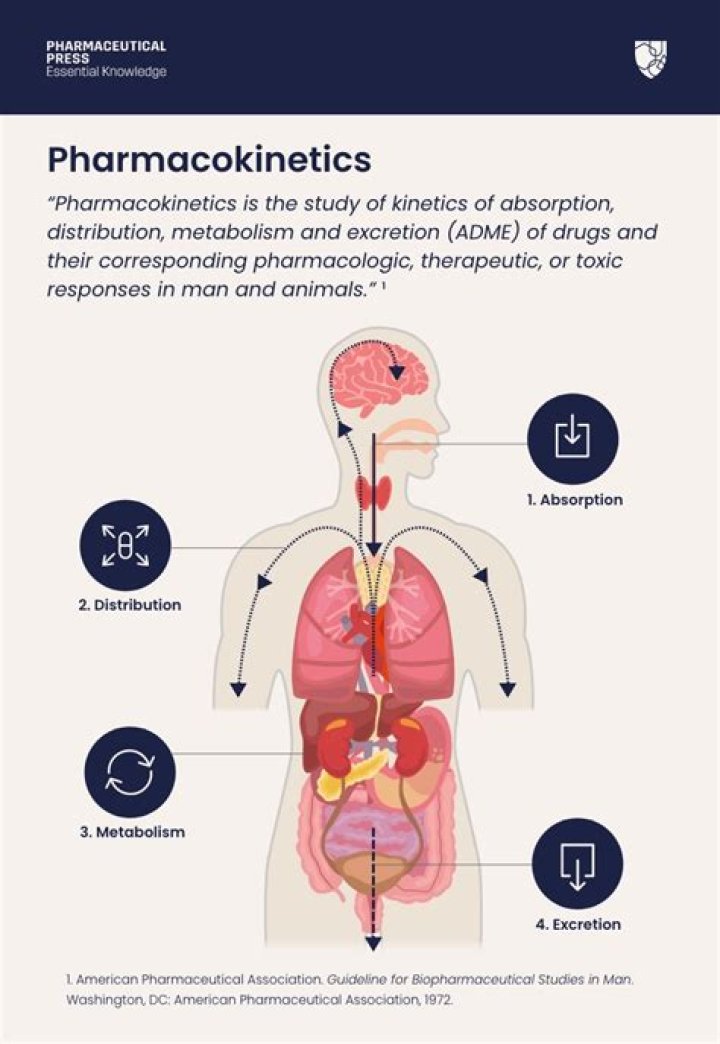
Hereof, what are pharmacokinetic studies?
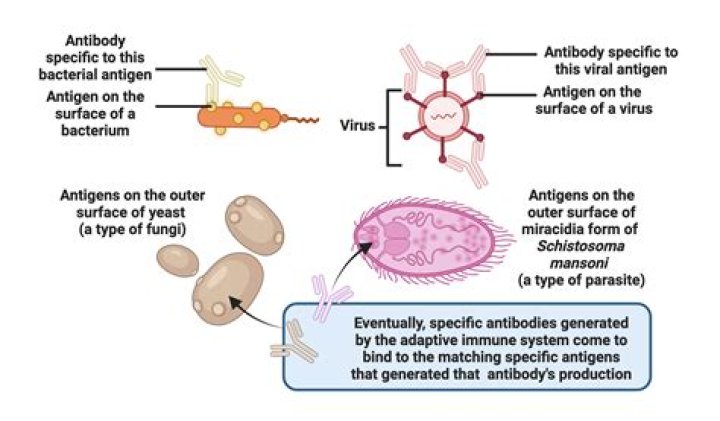
Pharmacokinetics Studies on the absorption, distribution, metabolism, and excretion of drugs and studies on the enzymes, transporters, etc. involved in these processes. In the case of human studies, the profile of a drug and its metabolites in blood; their half-lives and rates of elimination, for example, are studied.
Also Know, what are the 4 steps of pharmacokinetics? Pharmacokinetics involves four basic steps: absorption, distribution, metabolism, and excretion (ADME). Bioavailability of a drug, defined as the amount of drug that is actually available at the active site of receptor, is an important parameter of pharmacokinetics.
Keeping this in consideration, what is the purpose of a PK study?
A pharmacokinetic (PK) study of a new drug involves taking several blood samples over a period of time from study participants to determine how the body handles the substance. These studies provide critical information about new drugs.
What are 5 pharmacokinetic principles?
They are absorption, distribution, metabolism, and excretion.
Related Question Answers
How do you do Pharmacokinetic studies?
A standard pharmacokinetic study is the conventional method for evaluating the pharmacokinetics of a drug in human subjects. In such a study, subjects are given a single dose or repeated doses of an investigational drug. Then, blood and urine samples are collected in compliance with a fixed schedule.What is an example of pharmacodynamics?
Pharmacodynamics is the science or study of how the body reacts to drugs. An example of pharmacodynamics is someone studying how methadone affects a person getting over a heroin addiction.What the body does to a drug?
Pharmacokinetics, sometimes described as what the body does to a drug, refers to the movement of drug into, through, and out of the body—the time course of its absorption, bioavailability, distribution, metabolism, and excretion. Drug pharmacokinetics determines the onset, duration, and intensity of a drug's effect.Why is the study of pharmacokinetics so important?
Some of the most important pharmacokinetic properties to understand during pre-clinical testing include distribution, absorption, metabolism, and excretion. Pharmacokinetic testing can ensure that drugs do not fail during clinical trials for reasons that could have been predicted and avoided.What is the difference between pharmacodynamic and pharmacokinetic?
Pharmacokinetics (PK) refers to the movement of drugs through the body, whereas pharmacodynamics (PD) refers to the body's biological response to drugs. PK describes a drug's exposure by characterizing absorption, distribution, bioavailability, metabolism, and excretion as a function of time.What is a PK profile?
➢ Pharmacokinetics (PK) – the mathematics of the time course of Absorption, Distribution, Metabolism, and Excretion of drugs in the body. ➢ A favorable PK profile is vital to the therapeutic success of a drug. ➢ Drug must be able to reach its intended target.What is the purpose of pharmacodynamics?
Pharmacodynamics deals with the relationship between drug dosage or concentration in the body and its drug effects, both desirable and undesirable. Thus, it deals with the mechanism(s) of drug action and generally describes what a drug does to the body.What are pharmacokinetic parameters?
Pharmacokinetic parameters are assessed by monitoring variations in concentration of the drug and/or its metabolites in physiological fluids that are easy to access (i.e., plasma and urine). Plasma concentrations are usually checked, and in addition biopsies can be taken from animals and sometimes from humans.What is BA study?
About BA/BE Bioequivalence (BE): Bioequivalence of a drug product is achieved if its extent and rate of absorption are not statistically significantly different from those of the reference product when administered at the same molar dose.What does bioequivalent mean?
Bioequivalence is a term in pharmacokinetics used to assess the expected in vivo biological equivalence of two proprietary preparations of a drug. If two products are said to be bioequivalent it means that they would be expected to be, for all intents and purposes, the same.What is the steady state of a drug?
Steady-state concentration is the time during which the concentration of the drug in the body stays consistent. For most drugs, the time to reach steady state is four to five half-lives if the drug is given at regular intervals—no matter the number of doses, the dose size, or the dosing interval.What are the four major components of pharmacokinetics?
There are four main components of pharmacokinetics: liberation, absorption, distribution, metabolism and excretion (LADME). These are used to explain the various characteristics of different drugs in the body. They are covered in more detail below.What are the 3 phases of drug action?
A tablet or capsule taken by mouth goes through three phases—pharmaceutic, pharmacokinetic, and pharmacodynamic—as drug actions occur. In the pharmaceutic phase, the drug becomes a solution so that it can cross the biologic membrane.What is the ADME process?
ADME stands for absorption, distribution, metabolism, and excretion with respect to a specific topic in pharmacology known as pharmacokinetics. It isn't as hard as it all sounds. When you eat, you must absorb your food. Then this food must be sent all over the body, or distributed.What is an ADME study?
ADME studies are designed to investigate the absorption, distribution, metabolism and excretion of novel pharmaceuticals to support discovery, lead candidate selection, preclinical testing and clinical programs.Why is ADME important?
ADME studies provide important information regarding metabolism and excretion of compounds into urine and feces. The purpose of these studies is not only to understand the metabolism, but also how both parent and metabolites are eliminated. In addition, the excretion data also shed light on the role of transporters.What is a PK sample?
A pharmacokinetic (PK) study of a new drug involves taking several blood samples over a period of time from study participants to determine how the body handles the substance. These studies provide critical information about new drugs.What is half life of a drug?
The elimination half-life of a drug is a pharmacokinetic parameter that is defined as the time it takes for the concentration of the drug in the plasma or the total amount in the body to be reduced by 50%. In other words, after one half-life, the concentration of the drug in the body will be half of the starting dose.What are the 8 routes of drug administration?
Each route has specific purposes, advantages, and disadvantages.- Oral route. Many drugs can be administered orally as liquids, capsules, tablets, or chewable tablets.
- Injection routes.
- Sublingual and buccal routes.
- Rectal route.
- Vaginal route.
- Ocular route.
- Otic route.
- Nasal route.