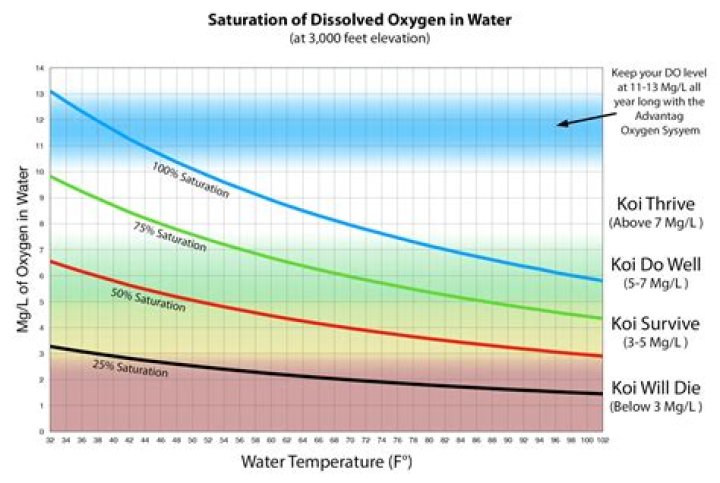
The primary cause of oxygen depletion in a water body is from excessive algae and phytoplankton growth driven by high levels of phosphorus and nitrogen. During the nighttime hours, these photosynthetic organisms consume oxygen through respiration when engaging in active photosynthesis. Hereof, how do you increase oxygen levels in water?
Simple aeration or agitation can increase dissolved oxygen enough to prevent problems. Injecting air or, especially, pure oxygen can increase levels as well, but only as high as saturation levels. Paying attention to temperature can also help improve DO, as colder water can hold more oxygen.
One may also ask, how do you fix dissolved oxygen in water? A sample bottle is filled completely with water (no air is left to skew the results). The dissolved oxygen in the sample is then "fixed" by adding a series of reagents that form an acid compound that is then titrated with a neutralizing compound that results in a color change.
Then, what affects dissolved oxygen levels in water?
Dissolved oxygen concentrations are constantly affected by diffusion and aeration, photosynthesis, respiration and decomposition. While water equilibrates toward 100% air saturation, dissolved oxygen levels will also fluctuate with temperature, salinity and pressure changes ³.
Why can cold water hold more oxygen?
Organisms typically have an optimum range in which they do best. The temperature and salinity of water influence how much oxygen it can hold. Warm water holds less dissolved oxygen than cold water because the molecules are moving faster than in cold water and thereby allow oxygen to escape from the water.
Related Question Answers
How do I get more oxygen in my soil?
Root Pruning Pots: In addition to root pruning and moisture control, fabric pots deliver plentiful oxygen to the root zone. For your growing plants, that's a breath of fresh air. Fabric pots are scientifically proven to create incredibly healthy roots. What causes low dissolved oxygen?
Low dissolved oxygen (DO) primarily results from excessive algae growth caused by phosphorus. This can result in insufficient amounts of dissolved oxygen available for fish and other aquatic life. Die-off and decomposition of submerged plants also contributes to low dissolved oxygen. How does temperature affect oxygen levels in water?
First, the solubility of oxygen decreases as temperature increases ¹. This means that warmer surface water requires less dissolved oxygen to reach 100% air saturation than does deeper, cooler water. Water at lower altitudes can hold more dissolved oxygen than water at higher altitudes. Does pH affect dissolved oxygen levels in water?
For example, dramatic temperature increases lowers the dissolved oxygen level. Also, if the pH is too low it decreases the ability of fish to effectively absorb dissolved oxygen. The optimal level for dissolved oxygen is 9.1 mg/L, and for pH is roughly 7.4. Can you add oxygen to water?
You can add oxygen to your tank by slowly pouring water into it from some height above. The water will pick up air en route as well as drive oxygen into the tank water. How much oxygen is added depends on how high above the tank you pour the water and how many times you repeat this procedure. How can oxygen levels be controlled?
How is the oxygen level in water controlled? - Place a paddle-wheel type device that mixes the water so that more water comes into contact with the air. This process is used in open pond systems.
- Force pressurized air (or pure oxygen) into the water with diffusers.
- Allow the water to free fall through the air.
How do you test oxygen levels in water?
Measuring dissolved oxygen in water. DO is measured using a dissolved oxygen meter (or DO meter). The best method is to take the measurement in the field at the same time each day, because DO concentrations can vary throughout a 24 hour period. What factors affect dissolved oxygen levels?
Dissolved oxygen concentrations are constantly affected by diffusion and aeration, photosynthesis, respiration and decomposition. While water equilibrates toward 100% air saturation, dissolved oxygen levels will also fluctuate with temperature, salinity and pressure changes ³. Why is dissolved oxygen in water important?
Dissolved oxygen (DO) is one of the most important indicators of water quality. It is essential for the survival of fish and other aquatic organisms. Oxygen dissolves in surface water due to the aerating action of winds. When dissolved oxygen becomes too low, fish and other aquatic organisms cannot survive. What happens if there is too much dissolved oxygen in water?
If too Much -- Total dissolved gas concentrations in water should not exceed 110 percent. Concentrations above this level can be harmful to aquatic life. The lower the concentration, the greater the stress. Oxygen levels that remain below 1-2 ppm for a few hours can result in large fish kills. What are the consequences of low dissolved oxygen in water?
The effect of low (>2 -4 mg/L) to hypoxic (>0 to 2.0 mg/L) dissolved oxygen concentrations upon fish and other aquatic life is highly dependent on the duration and extent of hypoxia, the sensitivity of individual species, their life stage and mobility, but it can cause acute stress and death (fish kills) or may impact What is it called when water loses its oxygen?
Aquatic hypoxia. Oxygen depletion is a phenomenon that occurs in aquatic environments as dissolved oxygen (DO; molecular oxygen dissolved in the water) becomes reduced in concentration to a point where it becomes detrimental to aquatic organisms living in the system. What is a good level of dissolved oxygen in water?
Healthy water should generally have dissolved oxygen concentrations above 6.5-8 mg/L and between about 80-120 %. What causes dissolved oxygen in water to increase?
Water with high concentrations of dissolved minerals such as salt will have a lower DO concentration than fresh water at the same temperature. Low dissolved oxygen (DO) primarily results from excessive algae growth caused by phosphorus. As the algae die and decompose, the process consumes dissolved oxygen. What is BOD water?
Biochemical oxygen demand (BOD) is the amount of dissolved oxygen needed (i.e. demanded) by aerobic biological organisms to break down organic material present in a given water sample at certain temperature over a specific time period. How do you check oxygen levels?
Share on Pinterest Blood oxygen levels may be measured using a pulse oximeter. The most efficient way to monitor blood oxygen levels is by an arterial blood gas or ABG test. For this test, a blood sample is taken from an artery, usually in the wrist. This procedure is very accurate, but it can be a little painful. What happens when dissolved oxygen is too low?
Low dissolved oxygen (DO) primarily results from excessive algae growth caused by phosphorus. As the algae die and decompose, the process consumes dissolved oxygen. This can result in insufficient amounts of dissolved oxygen available for fish and other aquatic life. Does dissolved oxygen change pH?
As the hydrogen ions increase, the water becomes more acidic; as the hydroxyl ions increase, the water becomes more basic. Acid rain and mining operations can lower the pH of water bodies. Dissolved Oxygen – Most aquatic organisms need oxygen to survive.  James Craig
James Craig