How many molecules are in c2h4?
 Michael Henderson
Michael Henderson 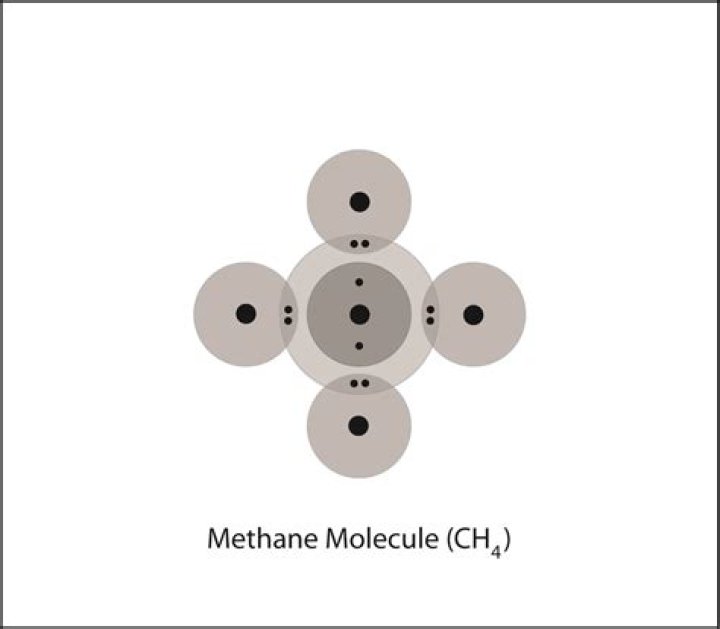
Keeping this in consideration, how many moles are in c2h4?
1 moles
Subsequently, question is, what is c2h4 classified as? Ethylene, C2H4, is a colorless gas. It is the simplest alkene. Because it contains a double bond, it it called an unsaturated hydrocarbon or an olefin. Ethylene is an odorless and colorless gas that is created by man-made sources.
Then, how many bonds does c2h4 have?
There are 5 sigma bonds (strong) and 1 pi bond (weak) in ethene. There are 4 (C-H) bonds (sigma) and 1 (C-C) bond (sigma). The valency of carbon is 4. Each carbon atom is bonded to 2 hydrogen atoms and there is a sigma bond between the two carbon atoms.
What is the percent by mass of carbon in c2h4?
Percent composition by element
| Element | Symbol | Mass Percent |
|---|---|---|
| Hydrogen | H | 14.372% |
| Carbon | C | 85.628% |
Related Question Answers
How many molecules are in a mole?
One mole (abbreviated mol) is equal to 6.022×1023 molecular entities (Avogadro's number), and each element has a different molar mass depending on the weight of 6.022×1023 of its atoms (1 mole).How many atoms are in 6.2 moles of aluminum?
How many atoms are in 6.2 moles of aluminum? I mol = 6.02X1023 atoms so 6.2 mol = 6.2x (6.02x1025) = 3.73 x1024 atoms of Al. 2.How do I calculate moles?
Use the molecular formula to find the molar mass; to obtain the number of moles, divide the mass of compound by the molar mass of the compound expressed in grams.What is the mass of 1 mole of nitrogen atoms?
14 gWhat is the atomic mass of ethane?
30.07 g/molWhat is molar mass equation?
Key Points. The molar mass is the mass of a given chemical element or chemical compound (g) divided by the amount of substance (mol). The molar mass of a compound can be calculated by adding the standard atomic masses (in g/mol) of the constituent atoms.What is the molar mass of Sulphur molecule?
256 gWhat is the Lewis dot structure of c2h4?
Drawing the Lewis Structure for C2H For C2H4 you have a total of 12 total valence electrons. Drawing the Lewis structure for C2H4 (named ethene) requires the use of a double bond. In a double bond two pairs of valence electrons are shared (for a total of four valence electrons).Is c2h4 a triple bond?
(A) The structural model for C2H4 consists of a double covalent bond between the two carbon atoms and single bonds to the hydrogen atoms. (B) Molecular model of C2H4. Triple bond in N2. Lewis structures can be drawn for molecules that share multiple pairs of electrons.Is c2h4 tetrahedral?
As a result they will be pushed down giving the C2H4 molecule a trigonal planar molecular geometry or shape with respect to the Carbon on the left. The C2H4 bond angle will be about 120 degrees since it has a trigonal planar molecular geometry.What bond is c2h4?
(A) The structural model for C2H4 consists of a double covalent bond between the two carbon atoms and single bonds to the hydrogen atoms. (B) Molecular model of C2H4. Triple bond in N2. Lewis structures can be drawn for molecules that share multiple pairs of electrons.Why is c2h4 sp2 hybridized?
The Lewis structure for etheneThe carbon atoms are sp2 hybridized. Two sp2 hybrids bond with the hydrogen atoms, and the other forms a sigma bond with the other carbon atom. The p-orbitals that are unused by the carbon atoms in the hybridizationoverlap to form the C=C.What is the chemical name for c2h4?
EtheneIs c2h4 a Lewis acid?
Answers : (4) C2H4 is CH2=CH2, it has one double bond that means it has pi electrons. It is electron rich . Therefore it is lewis base.Why is c2h2 a triple bond?
In drawing the Lewis structure for C2H2 (also called ethyne) you'll find that you don't have enough valence electrons available to satisfy the octet for each element (if you use only single bonds). The solution is to share three pairs of valence electrons and form a triple bond between the Carbon atoms in C2H2 .How many bonds are in c2h2?
The C2H2 molecule contains a triple bond between the two carbon atoms, one of which is a sigma bond, and two of which are pi bonds.Is c2h4 flammable?
Ethylene, C2H4, is a colorless flammable gas and is the simplest alkene. Ethylene, C2H4, is an unsaturated hydrocarbon and is a colorless flammable gas. It has a boiling point of -103.8C and a melting point of 169.4C. It is also used to make polyethylene.What is the chemical name for c2h6?
EthaneIs Ethyne and acetylene the same?
Acetylene (systematic name: ethyne) is the chemical compound with the formula C2H2. It is a hydrocarbon and the simplest alkyne. The carbon–carbon triple bond places all four atoms in the same straight line, with CCH bond angles of 180°.What is difference between ethene and ethylene?
Ethylene and ethene are one and the same compound, there is no difference between the two compounds. Ethylene is the common name of the compound while ethene is its IUPAC name. This hydrocarbon, ethene/ethylene is found in the group of hydrocarbons known as alkenes. These compounds have a carbon - carbon double bond.What is ch2ch2?
Ethene is a hydrocarbon which has the formula C2H4 or H2C=CH2. It is a colorless flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene (a hydrocarbon with carbon-carbon double bonds).What is the Colour of ethene?
The sample that decolourises (colour changes from brown to colourless) is ethene. This is because bromine attacks the double bonds in alkenes (electrophilic addition) to form bromoalkanes. The bromine is used up, thus the brown colour disappears.What is ethylene formula?
C2H4What is 3o2 in chemistry?
A photooxygenation is a light-induced oxidation reaction in which molecular oxygen is incorporated into the product(s).What is the formula of Methene?
The general formula for the alkenes is CnH2n. Note that there is no "Methene". Methene is not possible as alkenes require a carbon-carbon double bond and methane only has one carbon atom.What is the mass of c2h4?
28.05 g/molWhat is the mass percentage of C in ch3och3?
Elemental composition of CH3OCH3| Element | Symbol | Mass percent |
|---|---|---|
| Carbon | C | 52.1429 |
| Hydrogen | H | 13.1275 |
| Oxygen | O | 34.7296 |